PENUMBRA, noun.
1. (Astronomy) the partial or imperfect shadow outside the complete shadow of an opaque body, as a planet, where the light from the source of illumination is only partly cut off; the grayish marginal portion of a sunspot.
2. A shadowy, indefinite, or marginal area.
Steve Server // You are sitting at your computer in the neurosciences work room late one evening writing a progress note when your pager buzzes. A stroke code has been called in the Emergency Department. You grab your reflex hammer and your tuning fork and head down to the ED. The linoleum gives off a sterile glow as you walk, your instruments clinking with each step of your shoes. As you enter her room, the patient greets you. She looks comfortable. She is complaining of a mild headache, unsteadiness on her feet, and double vision.
And that’s it. No facial droop, no weakness, no tingling or numbness in any extremity. If you didn’t know any better, you may chalk this up to a migraine and send her on her way.
Except within neurology, things proceed in a methodical, regimented manner. The patient has earned herself a full neurological exam, which assesses her nervous system from head-to-toe:
Special tools and special techniques allow you to elicit abnormal findings. You do the exam the same way every time, so you never forget a step. After doing it so many times, the ritual comes quickly to you now. Your goal with the exam is to elicit positive findings, all in the hope of localizing a lesion—pinpointing the precise area of injury in the brain.
Your patient’s neurological exam is notable for only one pertinent positive finding. She has a right inferior homonymous quadrantanopia—an inability to see the lower right quadrant of her vision in both eyes. With that one clue, you have your localization: the superior left optic radiation. You wish her well as she is taken to the CT scanner so you can see whether your clinical localization correlates.
What can you do for her? Given that her symptoms began this morning, she is outside the 4.5 hour window to administer clot-dissolving medication to melt the blockage. All you can do now is protect the penumbra.
In a stroke, an area of brain dies due to lack of blood flow. This central infarction, however, is surrounded by a rim of brain tissue which has only been stunned and can be saved: this is the penumbra, and it needs to be saved at all costs. You’ll make sure that as much blood as possible can get past that blockage to save the narrow rim of brain which separates total disability from partial recovery.
The CT scan has been completed. You try to find an unoccupied computer somewhere. You’re starting to get a slight headache yourself. The light in the ED always bothers you—it is painfully white, but aggressively attenuated, a light you think must be similar to the light on a space station or in the underworld, emanating from some distant, unseen star.
Neurology is about being comfortable working in shade. There is a vast amount that we do not yet know about the brain. Consequently, there are not many therapies available to improve neurological outcomes, particularly involving diseases which directly damage neurons. Day-to-day, neurological rounds consist of visiting patients post-stroke who have residual deficits such as total unilateral paralysis, mutism, or the dreaded “locked in” syndrome, in which patients are totally paralyzed but for the ability to move their eyes up or down. Young patients with traumatic brain injury or multiple sclerosis. Patients with dementias, with ALS, with Creutzfeldt-Jakob Disease (often colloquially referred to as “Mad Cow Disease”)—relentlessly progressive diseases which are attended with grave prognoses and very few effective treatments. Bearing witness daily to neurological devastation, without the ability to offer much in the way of curative treatment, can be demoralizing.
But neurology as a discipline has evolved to thrive in the shadow of knowledge yet-to-be-known. From its birth, neurology has constructed itself as a field at the edge of understanding the brain. Until the mid-1800s, neurology and psychiatry were essentially the same field, concerned with behavior and sensation with little ability to correlate phenomena to specific sites of damage. By mid-century, however, neuropsychiatrists were eager to use brain dissection to find the site of disease. In this early period, seizure activity served as the critical focus for investigation on localization, and in the 1870s and 1880s, the likes of John Hughlings Jackson in England and Jean-Martin Charcot in France had carefully observed epileptic patients and offered their conclusions on the different sites in the brain responsible for speech, sensation, movement, etc. By 1884, the ability to correlate abnormal exam findings with lesions in the brain had developed enough to allow for the surgical resection of a brain tumor using only a small burr-hole over the area of predicted pathology (Kerr, Caputy, and Horowitz 1).
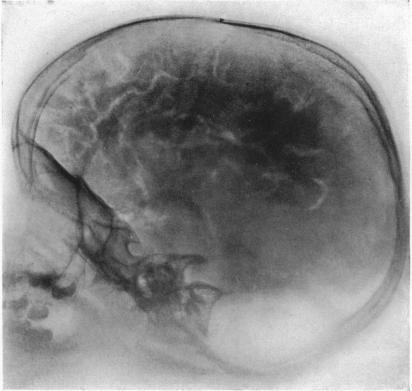
Before the advent of CT and MRI in the 1970s and 1980s, radiologic techniques for localization were invasive and uncomfortable: pneumoencephelography involved the draining of cerebrospinal fluid from the brain, and the injection of air or helium to fill the now-empty space in and around the brain. The patient was brought to get x-rays of the head, while moving their head in awkward orientations, often while suffering terrible headache and nausea. And even then, this modality could only ever offer a limited ability to see brain tumors. Good clinical examination still was necessary to localize sites of stroke, immunological lesion, or spinal cord pathology.
These days, with advanced imaging techniques of CT, MRI, and high-resolution angiography, neurology is less dependent upon an exam to offer insight into brain pathology. But nevertheless, the neurological exam retains an undeniable power in the day-to-day work of neurology.
In the face of so much unknown, is it any wonder that neurology remains so thoroughly committed to the special instruments and complex movements of the neurological physical exam? The exam is a ritual. As anthropologist Catherine Bell writes,
Ritualization tends to posit the existence of a type of authoritative reality that is seen to dictate the immediate situation. In many sociological analyses, this is one of the most basic social acts in the construction of reality (Bell 169).
The neurological exam is a catechism of the neurological world view, which demonstrates that by methodically probing and observing the body, we bring knowledge into being. Each time we examine a patient, we transform the disordered tangle of neurons within the skull into a system of positive and negative findings, diagnoses and prognoses.
On that foundation, we can build our plan to make a patient well. In neurology, that may be small changes over a long rehab process. The twitch of a toe. A thumbs-up. A look of recognition. A smile. A reaffirmed self-confidence. But with every exam, patients and doctors work together to shed light on the penumbra that is our neurological self.
Image: “Photograph of a roentgenogram.” From Walter E. Dandy, “Roentgenography of the brain after the injection of air into the spinal canal.” Ann. Surg. 70, 4 (1919): 397-403. Accessed https://commons.wikimedia.org/wiki/File:Pneumoencephalography.jpg.
Works Cited
Bell, Catherine. Ritual: Perspectives and Dimensions. New York: Oxford University Press, 2009.
Kerr, Caputy, and Horowitz. “A history of cerebral localization.” Neurosurg Focus 18, 4 (2005): E1.
UCL Clinical Skills. “The Full Neurologic Exam.” Youtube. https://www.youtube.com/watch?v=PEJJv3gJh74.